Blog
Ann Arbor Medical Malpractice Law Blog
Flint Motor Vehicle Accident Statistics
Flint is a great city with a lot going on, despite the negative attention and stereotypes placed on…
Delayed Cancer Diagnosis in Michigan: Common Fact Patterns That Create Liability
Very few types of medical malpractice have the same grave consequences as a delayed cancer diagnosis. In Michigan…
Hospital Communication Failures in Michigan: Consult Delays, Handoffs, and Missed Critical Labs
Hospital communication failures in Michigan aren’t just annoyances that lead to missed appointments, delays at the pharmacy, and…
OB/GYN Diagnosis Errors in Michigan: Ectopic Pregnancy, Preeclampsia, and Postpartum Hemorrhage
OB/GYN diagnosis errors in Michigan can range from routine misreadings to errors that lead to severe, and even…
Hypoxic-Ischemic Encephalopathy (HIE) and Cerebral Palsy Malpractice Cases in Michigan
Hypoxic-ischemic encephalopathy, or HIE, is a serious brain injury occurring in a fetus or newborn. It is caused…
Failure to Monitor Fetal Distress During Labor: Michigan Birth Injury Liability
The process of delivering a baby is naturally stressful for everyone involved. Parents, doctors, and nurses all have…
C-Section Delays and Fetal Distress Malpractice Claims in Michigan
Going to the hospital to bring a new child into the world should be an exciting, special time,…
Stroke Misdiagnosis and Delayed Treatment in Michigan Emergency Rooms
Suppose you went to a Michigan emergency room with symptoms of a stroke. They failed to diagnose you…
Hospitals Called Out for Unnecessary Back Surgeries on Seniors
The Lown Institute called out dozens of hospitals for performing unnecessary surgeries on Medicare beneficiaries with low back…
Radiology Errors and Missed Findings in Michigan: When a “Normal” Scan Is Malpractice
Radiology errors and missed findings in Michigan hospitals and clinics aren’t just annoyances that cause patients unnecessary return…
Expert Witness Requirements in Michigan Medical Malpractice Cases
If you have recently been injured or suffered a serious illness in the State of Michigan, and you…
Failure to Diagnose a Heart Attack in Michigan ERs: Red Flags and Legal Liability
Failure to diagnose a heart attack in Michigan ERs leads to tragic outcomes for patients and their families,…
Grand Rapids Motor Vehicle Accident Statistics
Grand Rapids, MI, is a thriving mid-sized city known for great breweries, renowned gardens, and multiple institutions of…
Kalamazoo County’s Most Dangerous Intersections
Kalamazoo is a vibrant Michigan city with a lot to love, from Western Michigan University and its beloved…
Michigan Car Accident Statistics – Traffic Fatalities MI
Michigan roadways are often beautiful, thanks to our state’s diverse geology, ecology, and history, but they can also…
Most Common Motor Vehicle Accident Injuries in Muskegon
Car crashes and associated injuries are an unfortunate reality throughout the modern world. It’s no surprise, then, that…
Filing a Wrongful Death Claim in an Ann Arbor Fatal Car Accident
Losing a loved one unexpectedly in a car crash is a devastating experience under any circumstances, but when…
Most Dangerous Roadways in Michigan: All You Need to Know
Michigan has beautiful woods, lakeshore scenery, and a claim to our nation’s automotive history, making it one of…
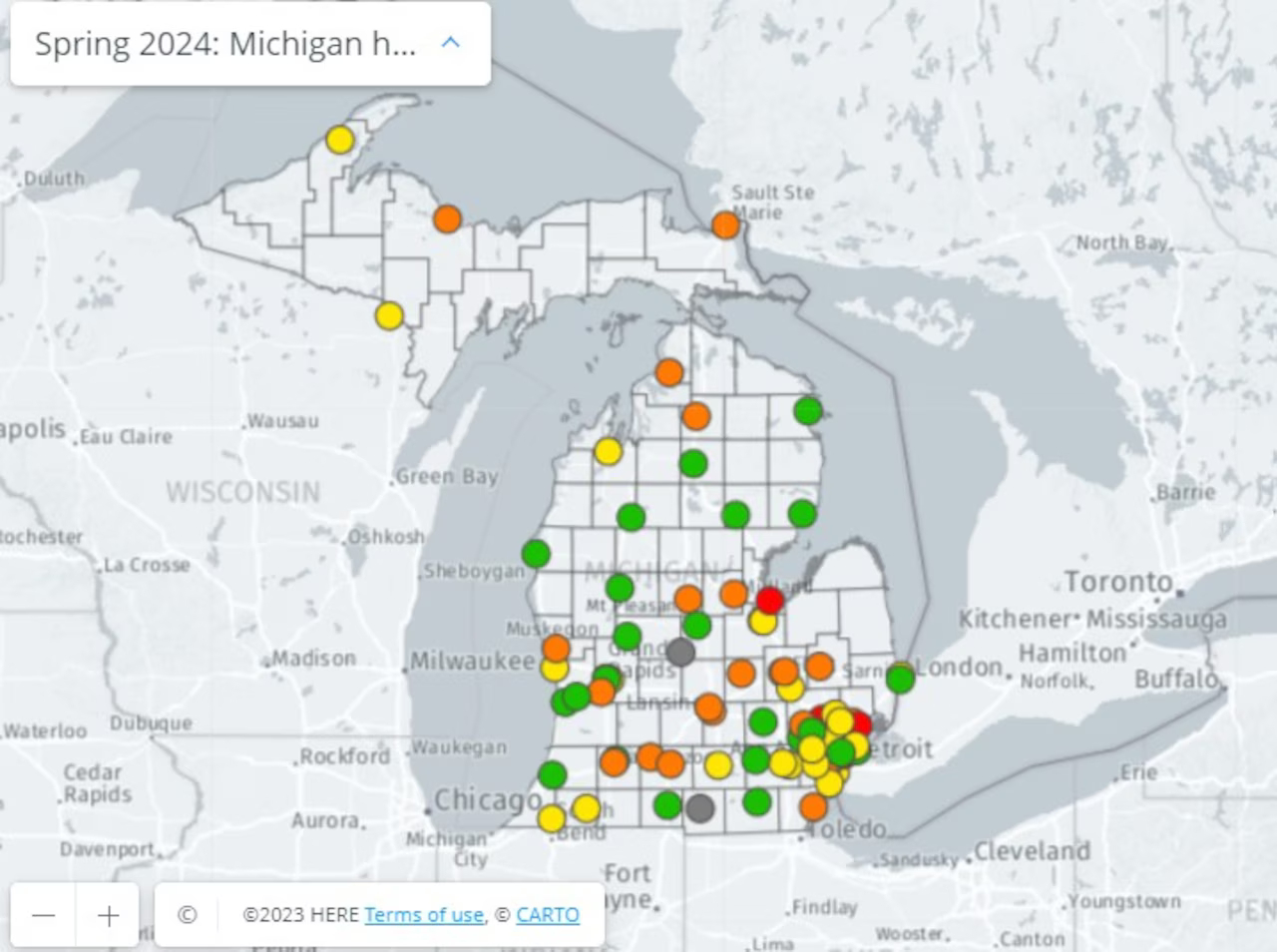
Safety Grades Declined for 16 Michigan Hospitals. How Did Yours Score?
Michigan’s spring 2024 hospital safety report card is here, with fewer “A” and “B” grades than the fall…
How Long Does a Medical Malpractice Lawsuit Take in Michigan?
Timing is often a pressing concern for individuals seeking justice and closure. The difficult answer is that they…
Goethel Engelhardt, PLLC
3049 Miller Road
Ann Arbor, MI 48103
Map